A quantum is a small, discrete amount of something. In physics, indivisible entities are often referred to as quanta. A photon, for example, is a quantum of light.
In science and engineering, mechanics is the set of rules for describing how things move. Quantum mechanics, then, is the set of rules for describing how quanta move.
The word quantum also refers to the discrete—or quantized—nature of some of the things we can measure, such as the energy of an electron in an atom.
Before we had quantum mechanics, we had what we now call classical mechanics. Newton’s laws are a great example of classical mechanics.
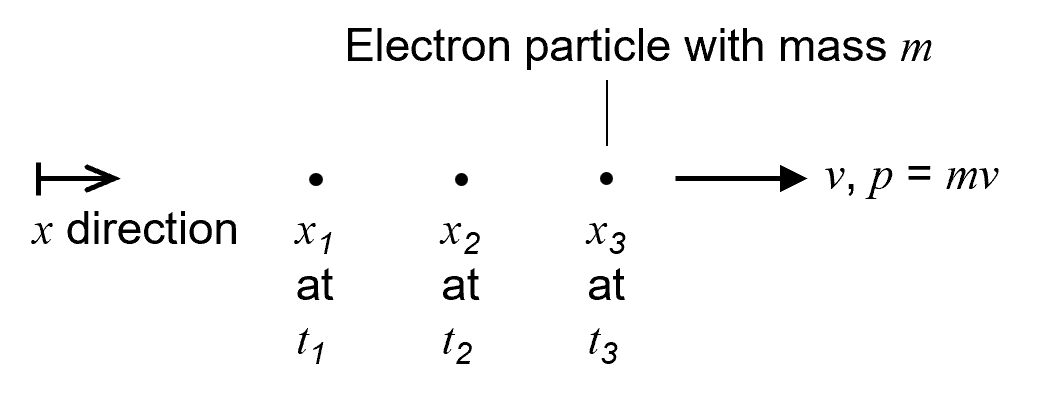
Imagine a particle with mass m. In classical mechanics, the defining aspects of the particle are its position x and momentum p. The momentum of the particle is its mass times its velocity v: p = mv. If we know a particle’s position and momentum, and the nature of any forces acting on it, then we can use the rules of classical mechanics to predict how the particle will move in space and time.
It turns out that classical mechanics does not work for quantum entities. We need quantum mechanics. At the macroscale, however, classical mechanics is an excellent approximation. We might as well use classical mechanics for macroscopic things like baseballs.
To get our foothold in quantum mechanics, we will focus on electrons. Here are some things to know about electrons:
- Electrons are one of the fundamental ingredients in the matter of the universe.
- Electrons have a negative electric charge. They are attracted to the positive electric charge of the protons in the nuclei of atoms.
- Electrons can be on their own, but they are usually found moving around the nuclei of atoms or moving between atoms.
What does an electron look like? In classical mechanics, electrons are particles. There is no reason to think of them as anything else. In quantum mechanics, the answer is not so simple. In fact, the answer is so complicated, uncertain, and foreign to our common sense that we will not get into it on this website. Instead, we will focus on understanding how electrons are measured to behave.
For the time being, we will just think of an electron as a thing with properties such as position and momentum that can be measured. Furthermore, we will focus on the state of the electron. Thinking in terms of states may be new for you. We will introduce the idea by talking about the state of an electron in classical mechanics.
Figure 1 shows an electron particle moving at constant velocity from left to right with no force acting on it. Its position is shown at three different times t. In classical mechanics, this is how we picture an electron moving along at constant velocity.