There are traditionally two main ways to perform calculations in quantum mechanics: matrix mechanics and wave mechanics. Both methods yield the same results. We will use wave mechanics here. In wave mechanics, the quantum state of an electron is represented by a wavefunction. The electron’s wavefunction tells us everything we can know about the electron. There is a law in quantum mechanics that governs how the wavefunction changes with time. We can query the wavefunction to find the probabilities of measuring various values of the position, momentum, and energy of the electron. We have a lot to learn!
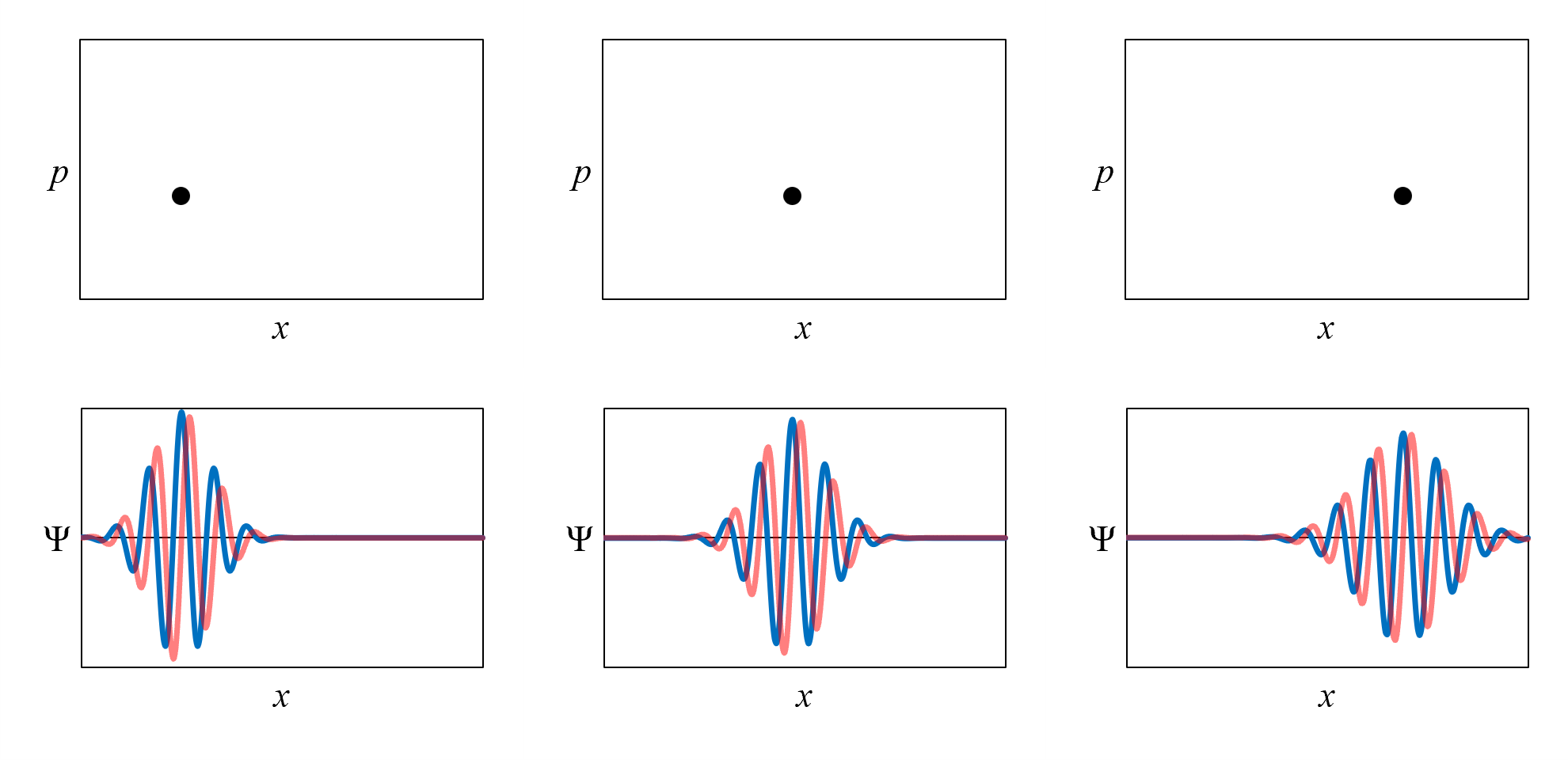
Figure 3, in the graphs on the top, shows the classical-state representation of an electron moving at constant velocity (and constant momentum) from left to right with no force acting on it. The classical state is shown at three times: t1 (left), t2 (center), and t3 (right). Remember, the classical-state representation is just an approximation.
The graphs on the bottom of Figure 3 show the quantum-state representation at the same three times. In the quantum-state representation, the electron is represented by a wavefunction Ψ. The wavefunctions shown are realistic snap-shot-in-time representations of an electron moving from left to right with no force acting on it.