In classical mechanics, electrons find themselves in classical states in which they have both definite positions and definite momenta. See, for example, Figure 2 in Chapter 1. The situation is different in quantum mechanics. An electron cannot be in a quantum state in which it has both a definite position and a definite momentum.
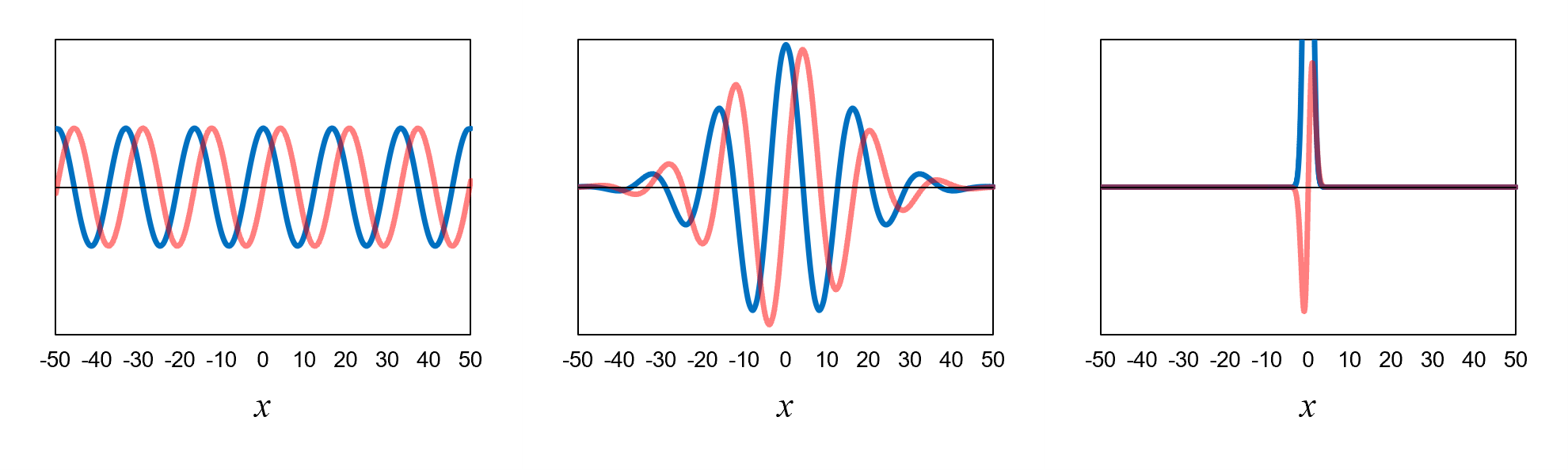
Consider an electron that has a definite momentum. It is represented by a wavefunction that is a definite-momentum eigenfunction such as the one shown in Figure 12, left. The function looks like this infinitely far in both directions. This is one end of the uncertainty principle spectrum: if an electron has an exact momentum, then it is in an equal superposition of all positions. We are entirely uncertain about its position.
In Chapter 5, we created a wavefunction for which the position of the electron was roughly contained within ±50. That wavefunction is reproduced in Figure 12, center. We did this by superposing eleven different definite-momentum eigenfunctions with momenta ranging from 1.9 to 6.1. The superposition resulted in destructive interference for positions less than −50 and greater than 50. Here we see that in order to reduce the uncertainty in position, we have to increase the uncertainty in momentum.
What if we want to know the position of the electron more precisely? For the wavefunction shown in Figure 12, right, the position of the electron is roughly contained within ±3.5. To make this happen, we have to superpose definite-momentum eigenfunctions with a range of momenta that is about 15 times the range of momenta used for the wavefunction in Figure 12, center. We see that in order to further reduce the uncertainty in position, we have to further increase the uncertainty in momentum.
To create a wavefunction representing an electron with a definite position (which would be a definite-position eigenfunction), we would have to superpose definite-momentum eigenfunctions including all values of momenta. This is the other end of the uncertainty principle spectrum.