So far, we have discussed an electron with no force acting on it. In the next two chapters, we will discuss an electron in a box and an electron in a hydrogen atom. These electrons feel forces.
In quantum mechanics, we do not explicitly specify the force acting on a quantum entity. Instead, we specify the distribution of potential energy, which implicitly describes the force. The relationship between force F and potential energy V is
F = −dV ∕ dx
Force is equal to how much the potential energy changes over a small increment of distance. The negative sign tells us the direction of the force: from high to low potential energy.
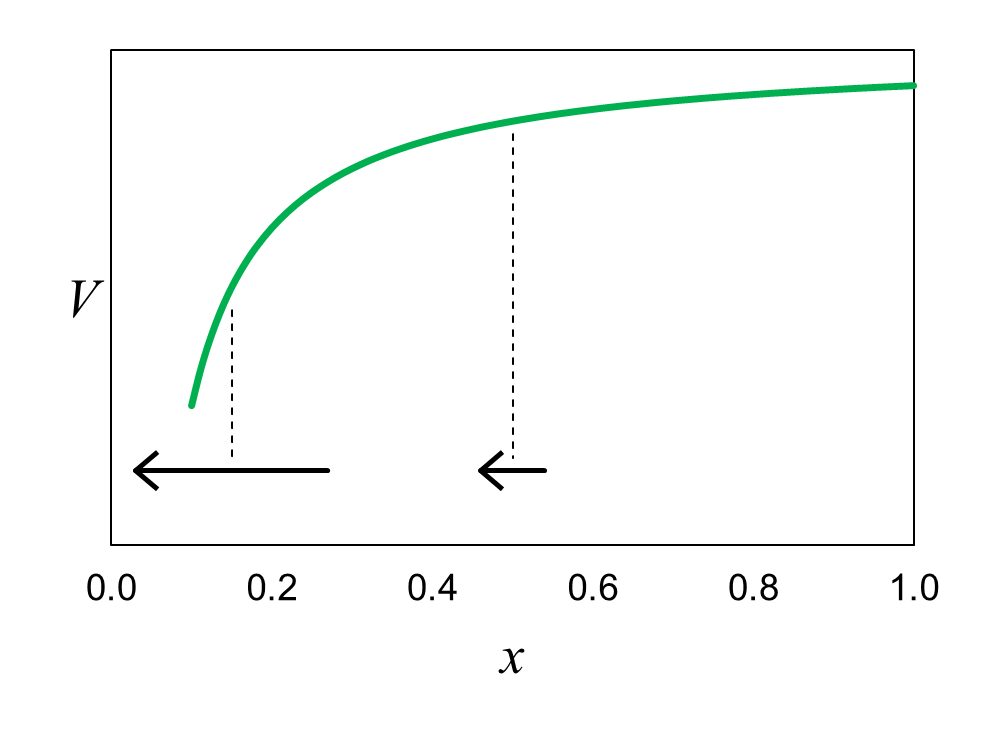
Figure 16 shows the distribution of electrostatic potential energy for an electron in the vicinity of a proton. The proton in this scenario is stationary at x = 0. If the electron is at x = 0.15, it feels a strong electrostatic force toward the proton (represented by the horizontal arrow) because the slope of the potential energy curve, dV/dx, is steep. If the electron is at x = 0.5, it feels a weaker force because the slope of the curve is more gradual.